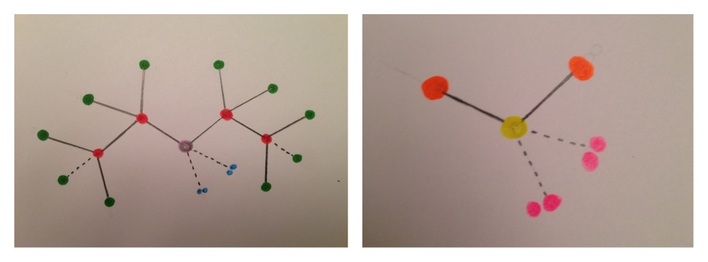
Color Key: (C4H10O) Color Key: (H2O)
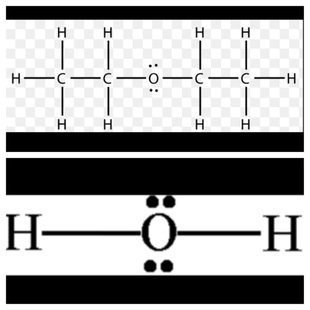
Oxygen Oxygen
Carbon Hydrogen
Hydrogen Unshared electrons
Unshared electrons
When C4H10O (diethyl ether) interacts with H2O (water), there are three intermolecular forces:
- Dipole-dipole: The positive H of H2O is attracted to the negative O of C4H10O.
- Hydrogen bonding: The H of C4H10 is attracted to the O of H2O.
- Dispersion Forces: These two molecules are adjacent in a liquid, so they are attracted by dispersion forces.
Below are the lewis structures for both molecules: (C4H10O and H2O)